Research
Significance
A central challenge in modern biomedical research is understanding how large, dynamic protein assemblies execute and regulate essential cellular processes—and how their dysfunction leads to human disease. The Takagi Lab addresses this challenge by applying single-particle cryo-electron microscopy (cryo-EM) to determine high-resolution structures of key proteins and protein complexes that govern transcription, metabolism, stress responses, immune signaling, and disease-relevant pathways.
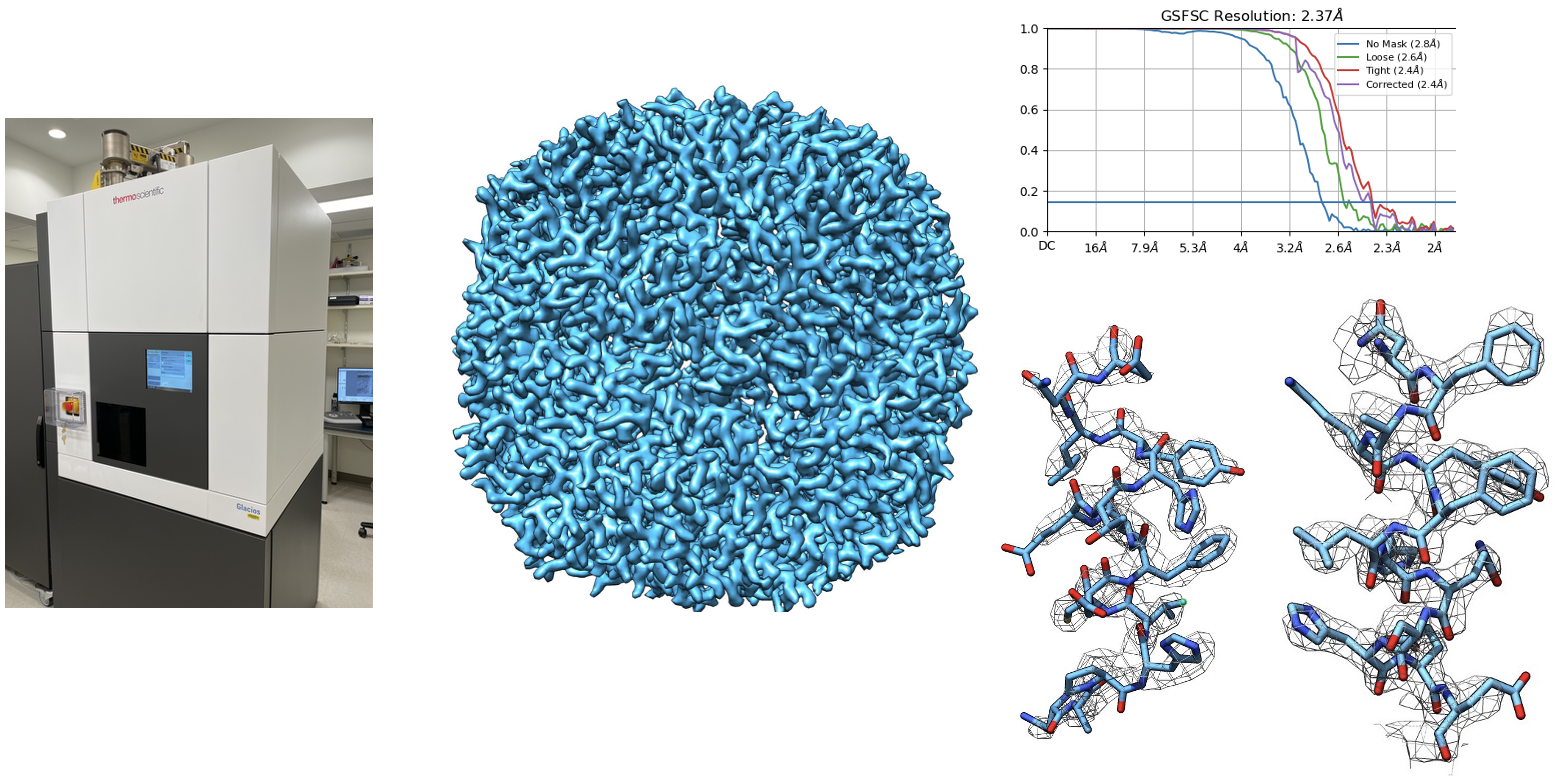
Originally focused on transcriptional regulation, the lab has expanded over the past seven years into a highly collaborative structural biology program, providing mechanistic insights across diverse biological systems. A unifying principle of our research is that direct visualization of protein structure and conformational dynamics is essential for understanding biological mechanism and for enabling rational therapeutic intervention.
Transcription and Gene Regulation
Precise control of transcription is fundamental to cellular identity, development, and homeostasis. Dysregulation of transcriptional machinery underlies numerous diseases, including cancer and developmental disorders.
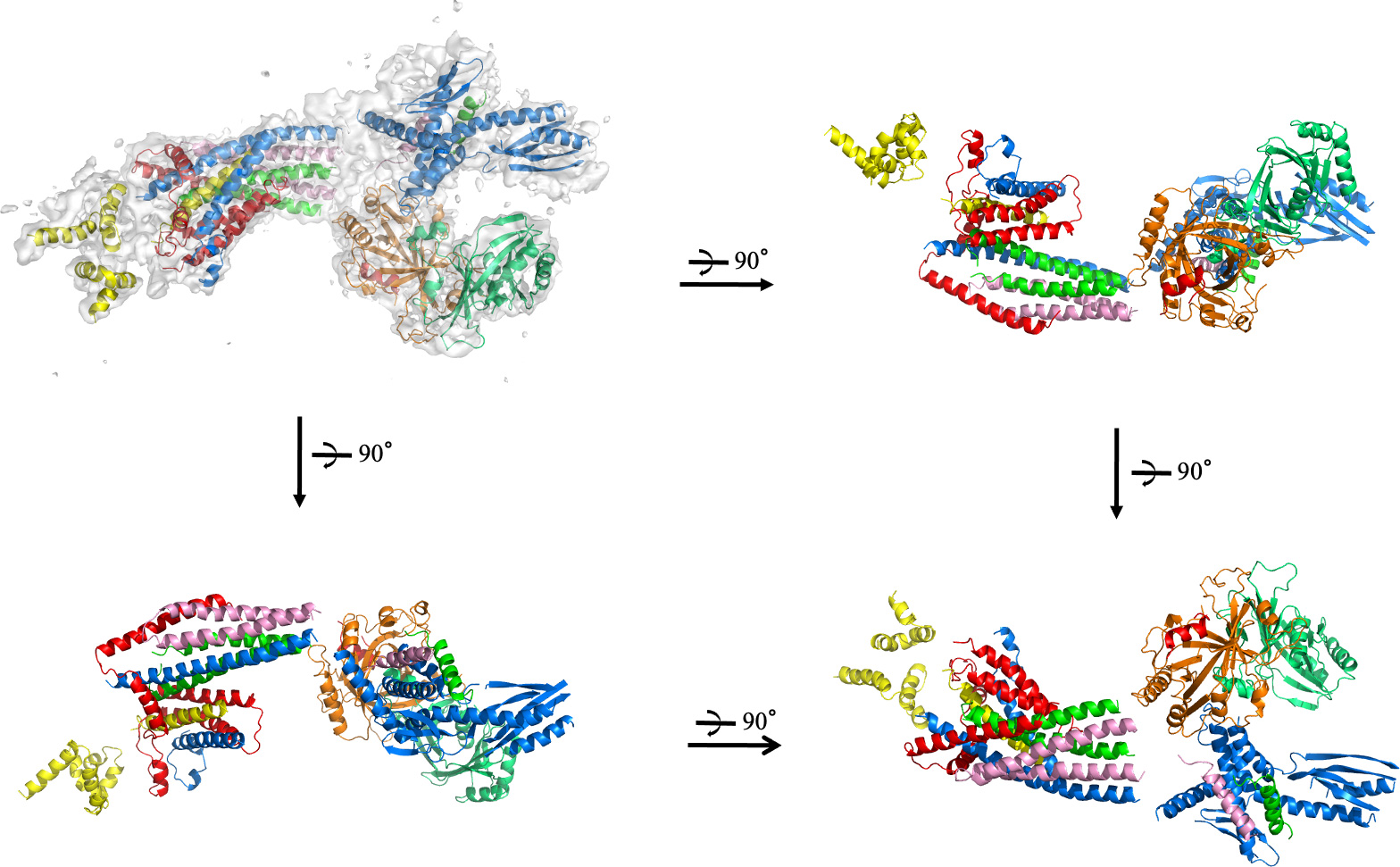
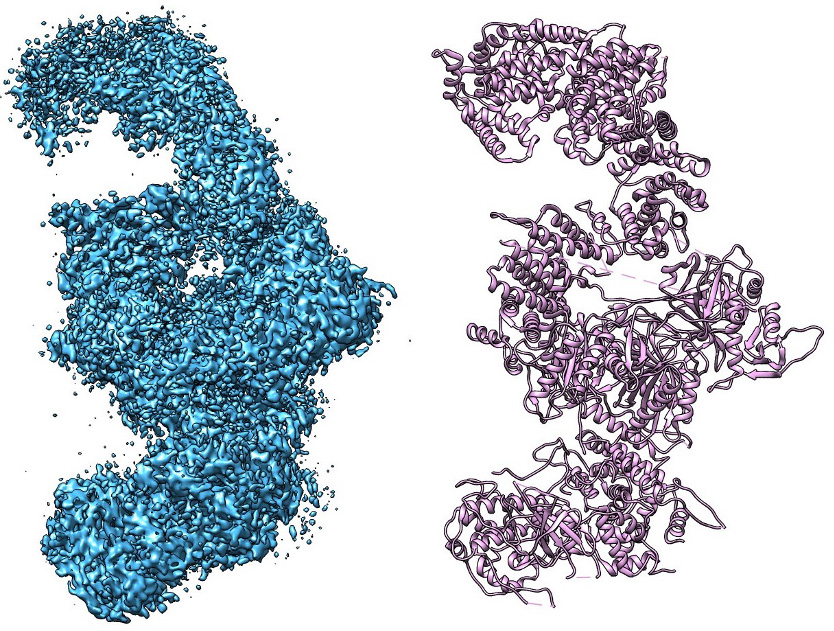
The Takagi Lab has a long-standing interest in transcription regulatory complexes, particularly the Mediator complex, which serves as a central integrator of regulatory signals between transcription factors and RNA polymerase II. By determining structures of transcription-related protein assemblies, our work provides critical insights into how regulatory information is transmitted at the molecular level and how alterations in complex architecture affect gene expression programs.
click to enlarge images:
Metabolism and Enzyme Mechanisms
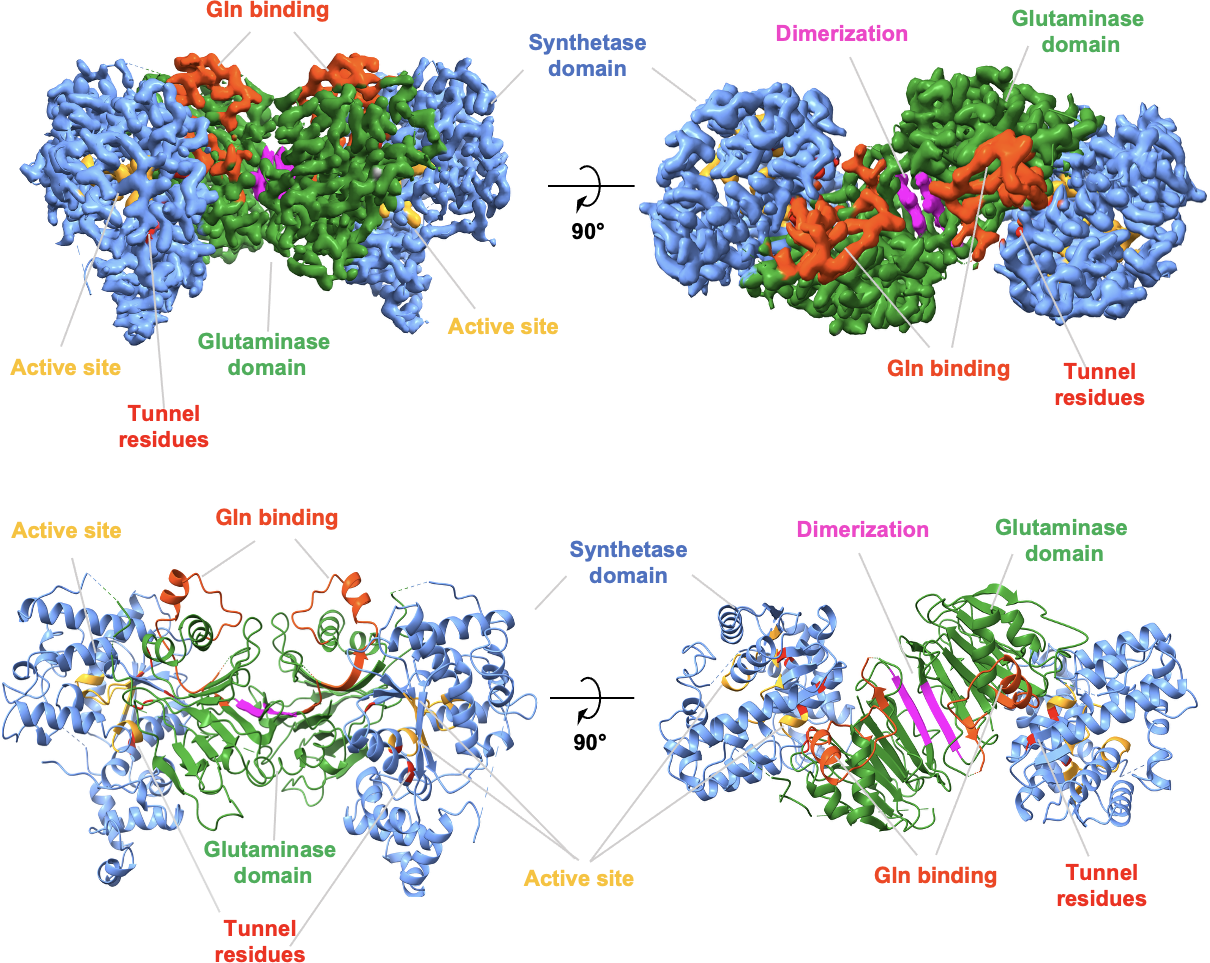
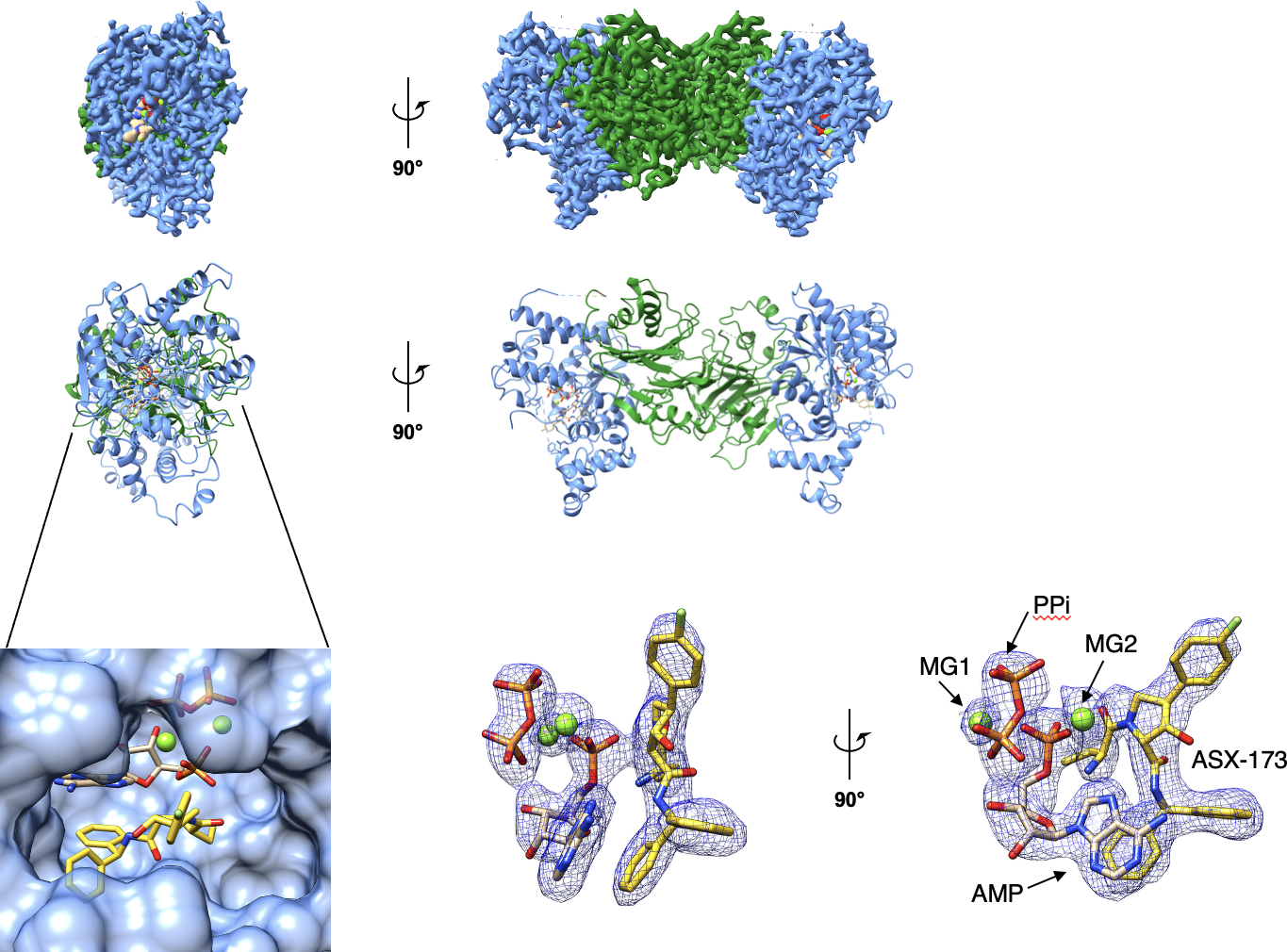
Metabolic enzymes play essential roles in sustaining cellular function, yet the molecular basis by which disease-associated mutations impair enzymatic activity often remains poorly understood. A major focus of the lab is human asparagine synthetase (ASNS), an essential enzyme implicated in neurodevelopmental disorders and cancer metabolism.
Our structural studies of ASNS define enzyme mechanism, conformational dynamics, and allosteric regulation, providing a foundation for structure-guided drug discovery.
click to enlarge images:
Translational Control and Stress Response Pathways
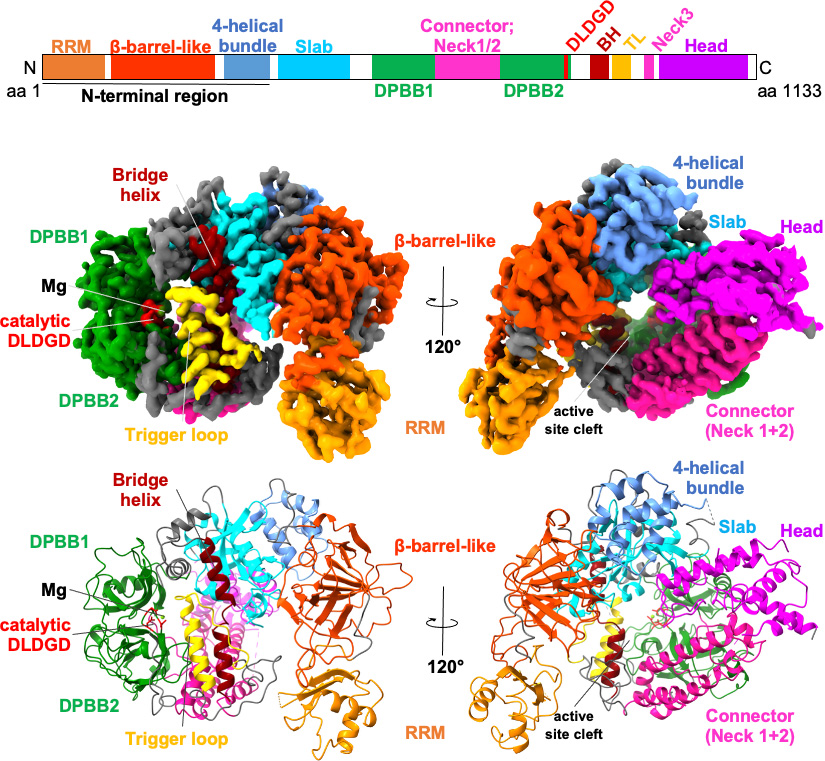
Cells rely on translational control mechanisms to adapt to nutrient limitation and stress. The GCN2-mediated integrated stress response (ISR) is a key pathway linking amino acid availability to global protein synthesis and cell fate decisions. The Takagi Lab applies cryo-EM to elucidate the structural basis of translational regulation within this pathway, revealing how large, dynamic protein assemblies sense stress and regulate translation initiation.
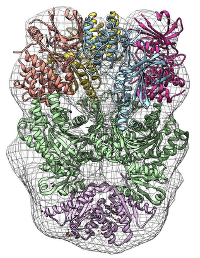
GCN2-mediated stress response pathway
Immunology and Cytokine Signaling
Cytokine signaling pathways orchestrate immune responses and inflammation. We investigate interleukin-9 (IL-9) signaling complexes to define receptor assembly, signaling specificity, and regulatory mechanisms.
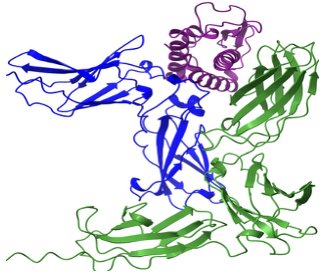
IL-9 signaling complexes
Metabolic Disease and Diabetes
We study insulin receptor–insulin analogue complexes to understand how ligand structure influences receptor activation and downstream signaling, informing the rational design of improved insulin analogues.
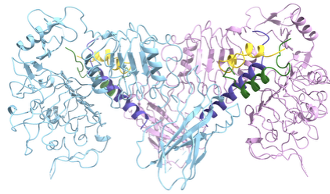
Insulin analogue-Insulin receptor
Technology Development and Innovation
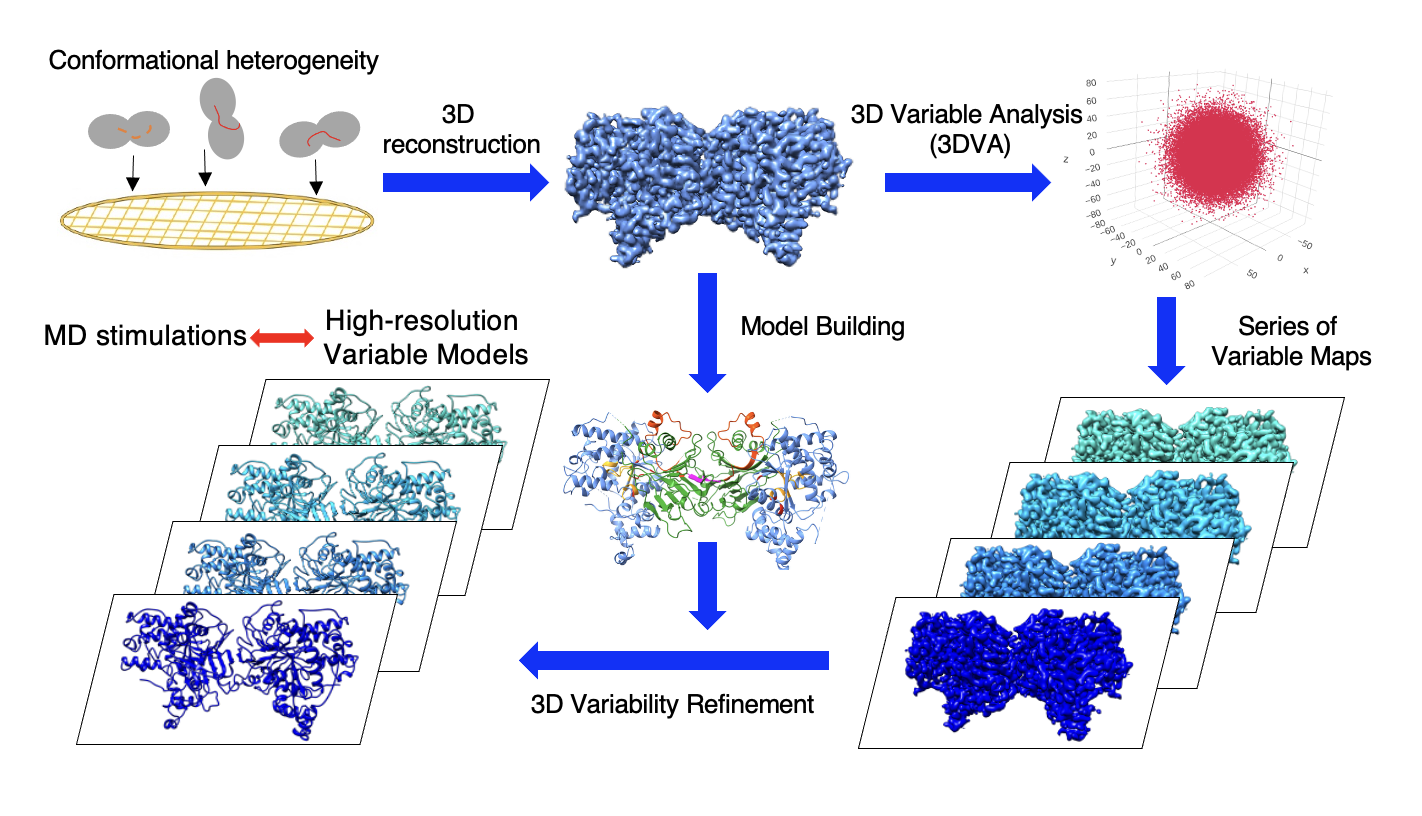
Our efforts include advanced protein complex engineering, cryo-EM methodologies for patient-derived biomarker structures, and computational tools to analyze protein motions and conformational heterogeneity.
Overall Impact
By determining structures and dynamics of key proteins and protein complexes, the Takagi Lab provides foundational knowledge that informs basic biology, therapeutic discovery, and translational medicine.
Current Research Funding